 For example, iron (II) sulphate (FeSO4) contains iron ions with an oxidation number of +2, whilst iron (III) sulphate (Fe2(SO4)3) contains iron ions with an oxidation number of +3. However, this only applies to positive oxidation states. If there is any risk of ambiguity, the specific oxidation number of an element in a given compound is shown using Roman numerals. 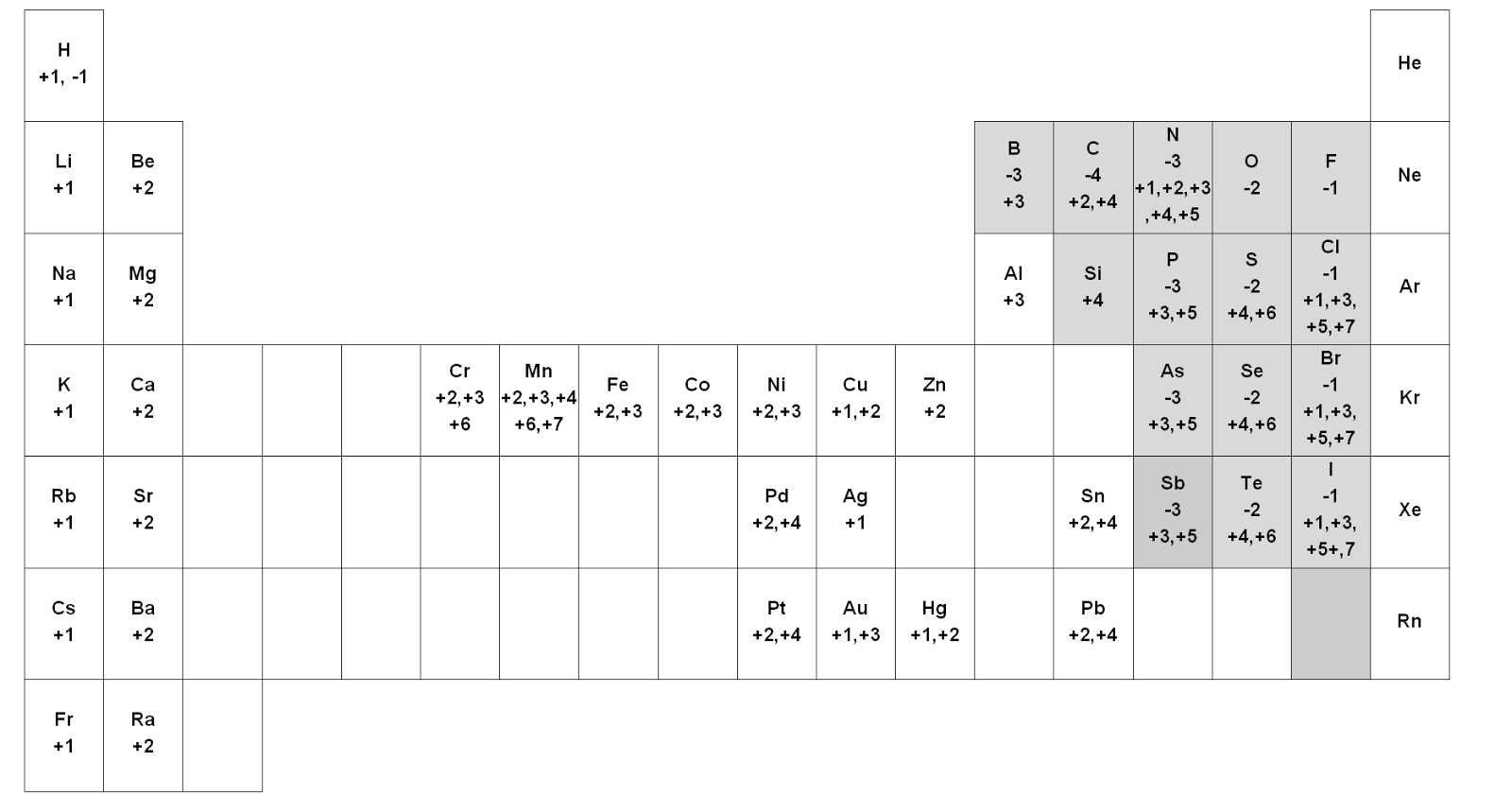
In fact, many elements can take numerous possible oxidation numbers, which can cause confusion in many compounds. Oxidation numbers and naming compoundsĪlthough we've just learned some rules for assigning oxidation numbers, they don't cover every element. This means that Cl must also have an oxidation number of +1, as 1 + 1 + (-2) = 0. 
H isn't in a metal hydride and so has an oxidation number of +1. For example, in HClO, O is the most electronegative element and so takes the most negative oxidation number. Once again, this is because oxygen and fluorine are more electronegative than chlorine. Likewise, chlorine takes variable oxygen numbers in compounds with oxygen or fluorine. We have two fluorines for every oxygen, and so the oxidation number of oxygen is +2. For example, in F2O, the more electronegative element is fluorine, so it gains the negative oxidation number -1. This is because we know that the more electronegative element takes the more negative oxidation number, and fluorine is more electronegative than oxygen. Oxygen also deviates from its usual oxidation number in compounds with fluorine. For example, in the case of H2O2, each hydrogen atom has the oxidation number +1, so each oxygen atom must have the oxidation number -1. Once again, this is a neutral compound, and therefore the sum of the oxidation numbers must be zero. But in peroxides, such as H2O2, it has an oxidation number of -1. Oxygen usually has an oxidation number of -2. For example, in NaH, Na has an oxidation state of +1 and H has an oxidation state of -1. This means that in a metal hydride, hydrogen must have an oxidation state of -1, as 1 + (-1) = 0. This is because we know that the sum of the oxidation numbers in a neutral compound is always 0, and that group 1 metals always have an oxidation number of +1. But in metal hydrides, such as NaH or KH, it has an oxidation number of -1. Hydrogen usually has an oxidation number of +1. Oxidation number exceptions As we've learned, there are a few exceptions to the oxidation numbers of elements within compounds. However, you must always remember the exceptions to the oxidation number rules. 
To help with working out the oxidation numbers of different compounds, here is an image of the periodic table with the common oxidation numbers per group.Ī periodic table with the oxidation numbers of the elements within their groups Group 1 elements all have an oxidation number of +1, Group 2 elements all have an oxidation number of +2, aluminium always has an oxidation number of +3, fluorine always has an oxidation number of -1, hydrogen usually has an oxidation number of +1 (except in metal hydrides), oxygen usually has an oxidation number of -2 (except in peroxides and in compounds with fluorine), and chlorine usually has an oxidation number of -1 (except in compounds with oxygen and fluorine). The more electronegative element in an ion or compound usually has the more negative oxidation number. The sum of the oxidation numbers in an ion equals the charge on the ion. The sum of the oxidation numbers of all the atoms or ions in a neutral compound equals 0. All uncombined elements have an oxidation number of 0, as they have neither lost nor gained electrons. 
There are a few rules that can help us work out oxidation numbers. A positive oxidation number shows that the element has lost electrons, while a negative oxidation number shows that it has gained electrons. So, what are oxidation numbers? Oxidation numbers are numbers assigned to ions that show how many electrons the ion has lost or gained, compared to the element in its uncombined state. Oxidation numbers are also used to name inorganic compounds. Oxidation numbers are important in chemistry because they help us keep track of the number of electrons transferred or shared during chemical reactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
